COVID-19 Testing and Good Microbiological Practice and Procedure

According to the Centers for Disease Control and Prevention (CDC), there are two kinds of tests available for COVID-19: viral test and antibody test. The viral or molecular test determines if a patient is actively infected with SARS-CoV-2, samples are collected through nasopharyngeal swabbing. On the other hand, the antibody or serology test also known as test for past infection, reveals if a patient has been exposed to the virus through blood samples. Most of the tests being rolled out against COVID-19 are viral tests based on reverse transcription. Collected samples should ideally be placed in a sterile transport tube immediately or on ice and special media to prevent degradation since the genetic material of SARS-CoV-2 is fragile and transient.
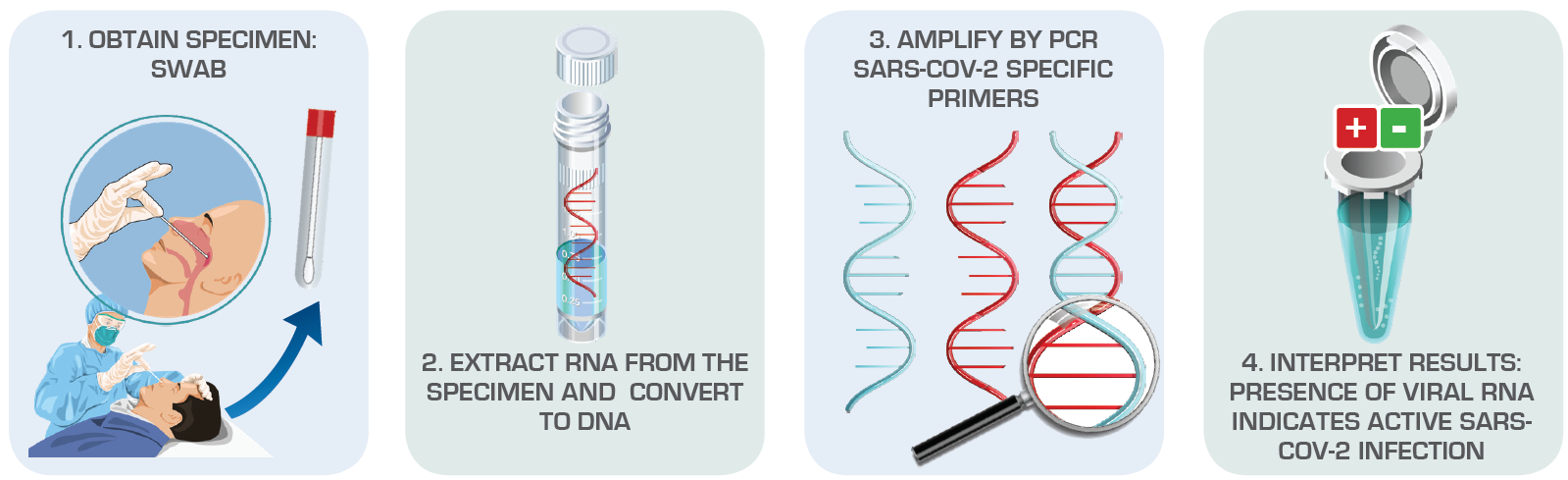
Figure 1. Viral Testing (Nucleic Acid Detection) Diagnose Active SARS-CoV-2 Infections Source: American Society for Microbiology
In contrast, the antibody test targets the immune response of infected person—detecting antibodies against the virus instead of detecting the viral genetic material. Although this test can give the result within an hour, most professionals claim that this type of test is not as accurate as viral testing. The problem is that antibodies only develop several weeks after an infection, which means that an antibody-based test is not recommended for early diagnosis and it might miss asymptomatic cases.
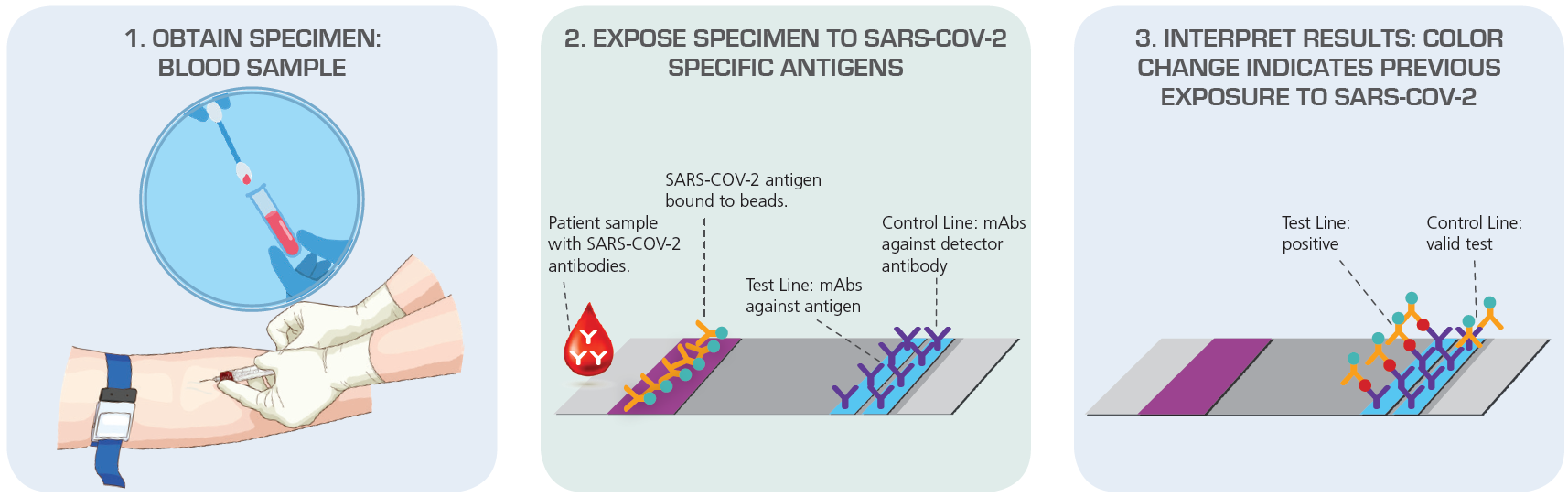
Figure 2. Antibody Test (Serology) Detect Immune Response to SARS-CoV-2 Exposure Source: American Society for Microbiology
Outlined below are the viral testing components for the CDC COVID-19 protocol “Centers for Disease Control and Prevention (CDC) 2019-Novel Coronavirus (2019-nCoV) Real-Time Reverse Transcriptase (RT)-PCR Diagnostic Panel:
Reagents and Supplies (Consumables)
Testing laboratories will need the following supplies dependent to the elements of the virus test kit provided by the CDC to run the COVID-19 test. To guarantee total compliance, secure that there will be no missing component.
- rRT-PCR primer/probe sets
- Positive template control
- RT-qPCR master mix
- Molecular grade water, nuclease-free
- Disposable powder-free gloves
- P2/P10, P200, and P1000 aerosol barrier tips
- Sterile, nuclease-free 1.5 mL micro centrifuge tubes
- 0.2 mL PCR reaction tube strips or 96-well realtime PCR reaction plates and optical 8-cap strips
- Laboratory marking pen
- Cooler racks for 1.5 micro centrifuge tubes and 96-well 0.2 mL PCR reaction tubes
- Racks for 1.5 ml micro centrifuge tubes
- Surface decontaminants
Equipment
Aside from the consumables, laboratories must also create an environment conducive to monitoring and carrying out the viral test. Laboratories that routinely conduct polymerase chain reactions likely conform to the environmental conditions. If not, the following equipment are essential for successful input in the coronavirus test.
- Vortex mixer
Efficiently mixes small volumes of liquids.
- Micropipettes (2 or 10 μl, 200 μl, and 1000 μl)
Adjustable calibrated instruments used with disposable sterile tips to transfer small quantities of liquid.
- Multichannel micropipettes (5-50 μl)
Adjustable calibrated instruments used with disposable sterile tips to transfer multiple samples of small quantities of liquid at a time.
- Cold blocks: 2 x 96-well cold blocks
Secure sample tubes and maintain a cool temperature while reagents and samples are added.
- Real-time PCR detection system
Combines adjustable automated PCR cycling with optical detection technology.

On top of the specified list of consumables and equipment required, CDC emphasized that viral isolation from specimens collected from people suspected to have COVID-19 should be performed by staff trained in the relevant technical and safety procedures in BSL-2 and BSL-3 laboratories. Otherwise, clinical laboratories should not attempt to conduct the viral isolation. World Health Organization (WHO) also added that national guidelines on laboratory biosafety should be followed in all circumstances. Corresponding to this, WHO released Laboratory Biosafety Guidelines for Handling and Processing Specimens Associated with COVID-19 and discoursed the following Good Microbiological Practice and Procedure (GMPP) as a core requirement in non-propagative diagnostic laboratory work (e.g. sequencing, NAAT).
Best Practices
- Do not store food or drink, or personal items like coats and bags in the lab. Eating, drinking, smoking, and/or applying cosmetics are also prohibited inside the laboratory.
- Do not put materials like gum and pens in the mouth while inside the lab.
- After handling any biological material, thoroughly wash hands preferably with warm running water and soap before leaving the laboratory.
- Do not place open flames or heat sources near flammable supplies and do not leave it unattended.
- Broken skin or any cuts should be covered prior to entering the laboratory.
- Supplies of lab equipment and consumables, including reagents, PPE, and disinfectants, should be sufficient and appropriate for the activities being executed prior to entry into the lab.
- To lessen the chance of accidents and incidents like spills, trips, or falls for lab personnel, supplies should be stored appropriately (per storage instructions) and safely.
- Proper labeling of all biological agents, chemicals, and radioactive materials should be followed.
- Barriers like plastic coverings should be used to protect written documents particularly those that may need to be removed from the laboratory.
- Work should be performed with care, in a timely manner, and without rushing. Avoid working when fatigued.
- Declutter materials unnecessary for the work procedure and keep the work area clean and tidy.
- Prohibit the use of earphones as it may distract personnel and halt equipment or facility alarms from being heard.
- Cover or remove any jewelry which may tear glove material, easily become contaminated or act as a catalyst for infection. Cleaning and decontamination of spectacles or jewelry should be considered if worn regularly.
- Avoid using mobile electronic devices especially when not required for the specific lab works or procedures being performed.
- Keep mobile electronic devices in areas where they could not easily become contaminated or act as a catalyst for infection. Use physical barriers or decontaminate the devices before leaving the lab if close proximity of such devices to biological agents is unavoidable.
Technical Procedures
- When manipulating specimens, use good techniques to minimize the formation of aerosols and avoid the inhalation of biological agents.
- Avoid the ingestion of biological agents and contact with skin and eyes.
- When handling specimens, do not forget to wear disposable gloves at all times.
- Contact of gloved hands with the face should be avoided.
- During operation, protect or shield the mouth, eyes, and face where splashes may occur.
- To prevent injury and injection of biological agents, handle all sharps and needles carefully.
- Replace any glassware with plasticware wherever possible.
- Use scissors with blunt or rounded ends in preference to those with pointed ends.
- Minimize the risk associated with the use of syringes or with needles and use ampoule openers for safe handling of ampoules.
- Never re-cap, clip or remove needles from disposable syringes.
- Puncture-proof or puncture-resistant containers fitted with sealed covers should be used in the disposal of any sharp materials like needles, needles combined with syringes, blades, and broken glass.
Preventing Dispersal of Biological Agents
- Leak-proof containers with lids properly secured should be used in the disposal of specimens and cultures before disposal in dedicated waste containers.
- Consider opening tubes with disinfectant soaked pad/gauze.
- At the end of the work procedures and if any material is spilled or contaminated, decontaminate work surfaces with a suitable disinfectant.
- For effective complete inactivation, the disinfectant should be efficient against the pathogen being handled and is left in contact with infectious waste materials for sufficient time.
When these practices are strictly implemented and the right laboratory equipment are used, the likelihood of exposure of personnel when handling or manipulating biological agents will be minimized.
References:
https://www.cdc.gov/coronavirus/2019-ncov/index.html
https://www.who.int/docs/defaultsource/coronaviruse/laboratory-biosafety-novel-coronavirus-version-1-1.pdf?sfvrsn=912a9847_2
https://asm.org/Articles/2020/April/COVID-19-Testing-FAQs